Preclinical Research
You are planning a preclinical in vivo study? We will give you the advice and support needed with our expertise and the highest technical standards in interventional procedures.
Our team consisting of radiologists, surgeons, scientists and qualified supporting personnel will guide the course of your study in a competent and professional way – from initiation until finalization.

Scientific basic research is the fundament of new applications, innovations and novel technologies – Together we will find answers and solutions!
More
Are you planning to proof the practicality of your proposed idea or to develop a prototype of a medical device? We will support you in implementing your ideas!
More
Evaluating the therapeutic effects and safety aspects of your medical devices or surgical procedures constitutes a key milestone of the development process. We would be pleased to set and reach this milestone together with you!
More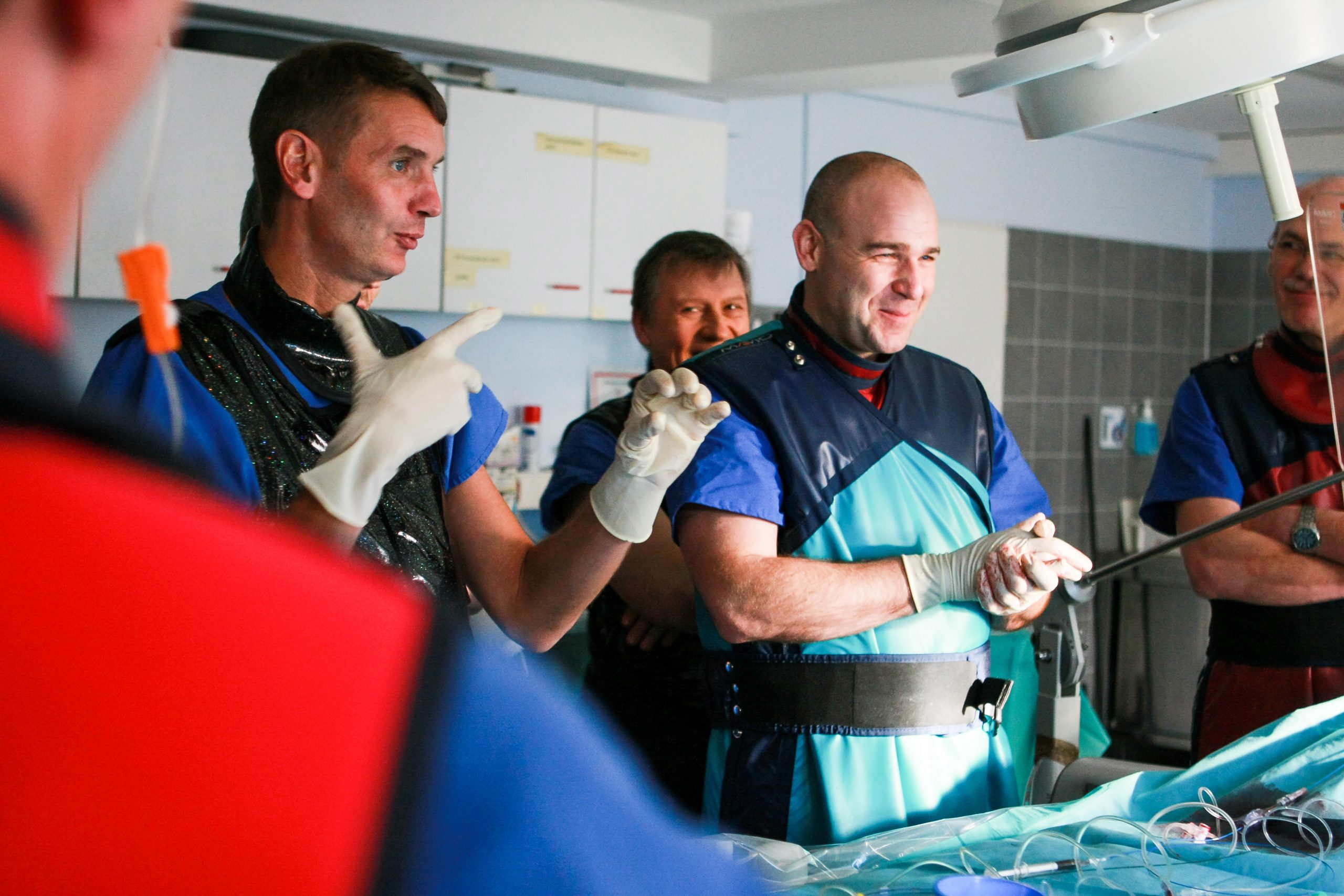
The clinical success of a medical device critically depends on the host response to the biomaterial composition. We can test the biocompatibility of your device in chronic long-term in vivo studies.
More